Alzheimer's Disease
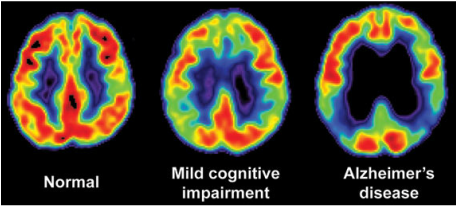
Ramifications on Health in the United States: By the year 2050, Alzheimer’s disease (AD) is predicted to affect 1 in 85 people worldwide (1). The United States spent about $203 billion in 2013 on health care related costs to AD, and that number is expected to rise to $1.2 trillion by 2050 (1). AD ranks in the top ten leading causes of death, but is the only disease in this category that cannot be prevented or slowed. While all other top ten diseases have seen a drop in the number of deaths since 2000, AD is the only disease that has seen a rise. In order to design treatments which can appropriately address AD there is a desperate need to have a fuller understanding for what drives the formation of AD at the molecular level.
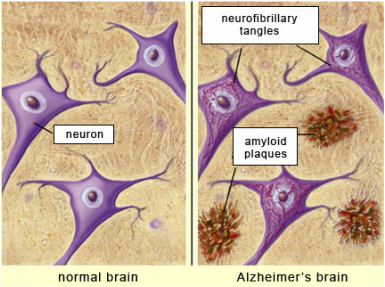
AD is the most common form of progressive senile dementia and is characterized by 1) deposition of a ~4 kDa peptide known as amyloid beta (Ab), which causes extracellular deposits of neuritic plaques and 2) paired helical filaments (PHFs) or single filaments (SFs) of the microtubule (MT) associated tau proteins, which form intracellular neurofibrillary tangles (NFTs) (2, 3) (see above figure). While the manifestation plaques and tangles are a hallmark of AD, the accumulation of NFTs inside neurons tracks closely with neuronal loss and cognitive function (4, 5). There is much debate concerning why this relationship exists between NFTs and these physiological events (6). Nonetheless, stereotypical patterns of NFT accumulation have been identified and form the basis for six pathological stages used to classify AD. The importance of NFT formation by tau is underscored by measures that are used to categorize postmortem neuropathological assessment of AD (7). The Masterson laboratory uses biophysical approaches to characterize the interaction of tau with a variety of cellular components that may lead to its misfolding. Students are able to apply techniques in enzymology, structural characterization through CD and FTIR spectroscopy, isothermal titration calorimetry, and NMR spectroscopy.
1. Alzheimer’s Organization website: http://www.alz.org/. Accessed June 24th, 2013.
2. Sambamurti, K.; Greig, N. H.; Lahiri, D. K. Advances in the cellular and molecular biology of the beta-amyloid protein in Alzheimer's disease. Neuromolecular Med. 2002, 1, 1-31.
3. Tolnay, M.; Probst, A. REVIEW: tau protein pathology in Alzheimer's disease and related disorders. Neuropathol. Appl. Neurobiol. 1999, 25, 171-187.
4. Chiba, T. In Emerging Therapeutic Strategies in Alzheimer's Disease, Neurodegenerative Diseases; Neurodegenerative Diseases; Intech: 2013; pp 182.
5. Perrin, R. J.; Fagan, A. M.; Holtzman, D. M. Multimodal techniques for diagnosis and prognosis of Alzheimer's disease. Nature 2009, 461, 916-922.
6. Crespo-Biel, N.; Theunis, C.; Van Leuven, F. Protein tau: prime cause of synaptic and neuronal degeneration in Alzheimer's disease. Int. J. Alzheimers Dis. 2012, 2012, 251426.
7. Braak, H.; Braak, E. Frequency of stages of Alzheimer-related lesions in different age categories. Neurobiol. Aging 1997, 18, 351-357.
AD is the most common form of progressive senile dementia and is characterized by 1) deposition of a ~4 kDa peptide known as amyloid beta (Ab), which causes extracellular deposits of neuritic plaques and 2) paired helical filaments (PHFs) or single filaments (SFs) of the microtubule (MT) associated tau proteins, which form intracellular neurofibrillary tangles (NFTs) (2, 3) (see above figure). While the manifestation plaques and tangles are a hallmark of AD, the accumulation of NFTs inside neurons tracks closely with neuronal loss and cognitive function (4, 5). There is much debate concerning why this relationship exists between NFTs and these physiological events (6). Nonetheless, stereotypical patterns of NFT accumulation have been identified and form the basis for six pathological stages used to classify AD. The importance of NFT formation by tau is underscored by measures that are used to categorize postmortem neuropathological assessment of AD (7). The Masterson laboratory uses biophysical approaches to characterize the interaction of tau with a variety of cellular components that may lead to its misfolding. Students are able to apply techniques in enzymology, structural characterization through CD and FTIR spectroscopy, isothermal titration calorimetry, and NMR spectroscopy.
1. Alzheimer’s Organization website: http://www.alz.org/. Accessed June 24th, 2013.
2. Sambamurti, K.; Greig, N. H.; Lahiri, D. K. Advances in the cellular and molecular biology of the beta-amyloid protein in Alzheimer's disease. Neuromolecular Med. 2002, 1, 1-31.
3. Tolnay, M.; Probst, A. REVIEW: tau protein pathology in Alzheimer's disease and related disorders. Neuropathol. Appl. Neurobiol. 1999, 25, 171-187.
4. Chiba, T. In Emerging Therapeutic Strategies in Alzheimer's Disease, Neurodegenerative Diseases; Neurodegenerative Diseases; Intech: 2013; pp 182.
5. Perrin, R. J.; Fagan, A. M.; Holtzman, D. M. Multimodal techniques for diagnosis and prognosis of Alzheimer's disease. Nature 2009, 461, 916-922.
6. Crespo-Biel, N.; Theunis, C.; Van Leuven, F. Protein tau: prime cause of synaptic and neuronal degeneration in Alzheimer's disease. Int. J. Alzheimers Dis. 2012, 2012, 251426.
7. Braak, H.; Braak, E. Frequency of stages of Alzheimer-related lesions in different age categories. Neurobiol. Aging 1997, 18, 351-357.